CADENCE Study
Multi-Cancer Screening Test (MCST) Development
Towards Cost-Effective Multi-Cancer Early Detection

Treatment of cancer is most successful and affordable when it is detected early.
With the rising cancer cases globally over the years, we have committed ourselves to finding an accessible and affordable solution in the form of a singular screening test capable of detecting multiple major cancers in their early stages.
Starting from Singapore, we have set eyes on expanding our scope with future regional, and eventually global studies in our quest for cost-effective early cancer detection.
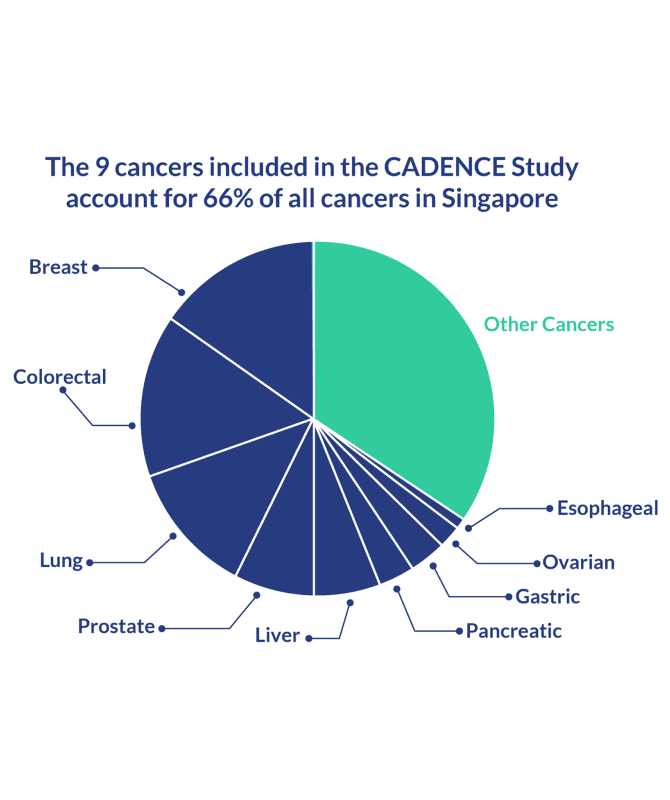
Cancer in Singapore
Cancer cases have been on the gradual rise in Singapore. According to the Singapore Cancer Registry, the cumulative incidence rate of cancer for the five-year period of 2013–2017 is 365.1 per 100,000 population, 19.6% more than the previous period.1
At this rate and with the current high prices of cancer drugs, Singapore’s expenditure on cancer drugs is expected to increase seven-fold by 2030.2
The steady increase in cancer burden is also shared by countries worldwide. GLOBOCAN 2020 reports that at the current rate, new cancer cases globally will increase by 47% by 2040.3
Currently, Singapore only has screening recommendations for breast, cervical and colorectal cancers, partly because of a lack of cancer screening tests with acceptable sensitivity, specificity and economic efficiency. As Singapore focuses on preventive healthcare to reduce healthcare costs and the burden of patients’ families, there will increasingly be a need for screening tests that can detect diseases such as cancer early, when they can be treated successfully.

A microRNA-centred multi-modal approach
MicroRNAs are short, non-coding nucleic acid strands that regulate gene expression. We had developed and commercialized GASTROClear, the world’s first microRNA-based blood test for the early detection of gastric cancer. Current research on microRNAs have also established them as highly sensitive biomarkers for other cancers.
However, other biomarkers (based on metabolites, proteins, RNA, and DNA) have also been shown to be promising for the early detection of cancer. In the CADENCE study, we will use an approach that focuses on microRNA biomarkers and their synergy with other biomarkers in achieving highly sensitive and specific cancer detection performance.
Our Study
In collaboration with key clinical experts and institutions in Singapore, we are embarking on the CADENCE (CAncer Detected Early caN be CurEd) study to develop a blood-based multi-cancer screening test (MCST) to detect the nine most prevalent cancers in Singapore: lung, breast, colorectal, liver, stomach, esophageal, ovarian, pancreatic, and prostate.
The CADENCE study is a 3-year cancer detection study investigating microRNA expression in tandem with other biomarkers such as DNA methylation patterns on a multi-centre cohort of over 20,000 participants – the largest cancer detection study regionally to date and the first of its kind.
CADENCE (CAncer Detected Early caN be CurEd) Study Pipeline
Target Selection
Discovery of potential microRNA biomarker candidates for all nine cancers
Biomarker Verification
Verification of candidate biomarkers, and refinement of the list of candidates to be used in assay development
Assay Development
Designing, building and finalization of the assay panels and algorithms
Clinical Validation
Evaluation of assay performance in a validation cohort
CADENCE MCST Team

- Lead Principal Investigator
- Chief Executive, National University Health System Senior Vice President (Health Affairs), National University of Singapore

- Dean, Lee Kong Chian School of Medicine Senior Vice President (Health & Life Sciences), Nanyang Technological University

- Executive Director, Singapore Translational Cancer Consortium Director, National University Cancer Institute, Singapore Deputy Director, Cancer Science Institute of Singapore

- Research Director, Cancer Translational Research Programme, NUS Yong Loo Lin School of Medicine Chief Scientific Advisor, Mirxes

- Co-founder and CEO, Mirxes
1 National Registry of Diseases Office. Singapore Cancer Registry Annual Report 2019.
Available at https://www.nrdo.gov.sg/docs/librariesprovider3/default-document-library/scr-2019_annual-report_final.pdf?sfvrsn=fa847590_0. Accessed Apr 2022.
2 The Straits Times. Why S’pore spends so much on cancer treatment and how MediShield Life changes aim to tackle this.
Available at https://www.straitstimes.com/singapore/health/why-singapore-spends-so-much-on-cancer-treatment-and-how-changes-to-medishield-life. Accessed Apr 2022.
3 Sung H et al. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA: Cancer J Clin 2021;71(3):209–249